Supplier:
BioLegendCat no: 580706
Recombinant Human TGF-beta1 (carrier-free)
Prices direct from BioLegend
Quick response times
Exclusive Absave savings/discounts
SPECIFICATIONS
Catalog Number
580706
Size
100 microg
Gene Id
7040
Storage Temp
-20 C or -70 C
Shipping Temp
Blue Ice
Storage Buffer
Unopened vial can be stored at 4C for three months or -20 to -70C for six months. For maximum results, quick spin vial prior to opening. Stock solutions should be prepared at no less than 10 microg/mL in buffer containing carrier protein such as 1% BSA or HSA or 10% FBS. For long term-storage, aliquot into polypropylene vials and store in a manual defrost freezer. Avoid repeated freeze/thaw cycles.
Alternative Names
Transforming growth factor beta 1
Regulatory Status
RUO
SUPPLIER INFO
Email:
info@biolegend.comMore from BioLegend
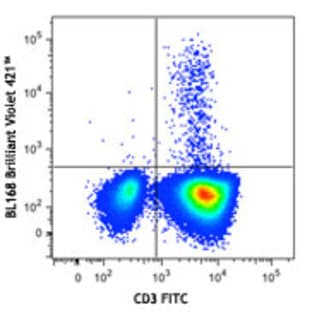
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
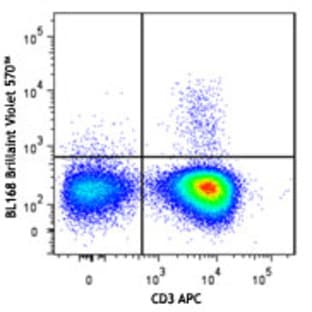
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
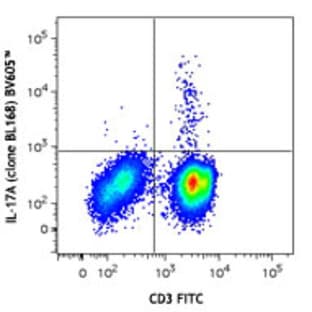
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
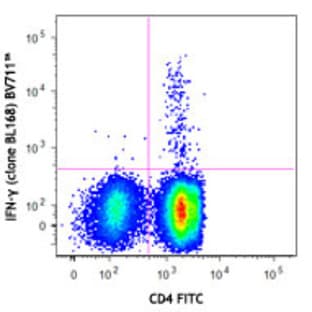
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
Applications
FC, ICC
Hosts
Mouse
Reactivities
Hum
Latest promotions
Buy any polyclonal or monoclonal antibody from our extensive range of pre-made antibodies and for a limited time only receive a $50 discount!(T&C apply:...
New brilliant antibodies, and new lower prices!For flow cytometry reagents in general, \"bright is better.\" The violet-excitable BD Horizon™ BV421 and...
Did your supplier increase the price of Fetal Bovine Serum? Did they substitute the US Origin with USDA? Well say no more! Innovative Research is still...
We're so sure that you'll prefer Cayman Assay kits over your present brand that we're willing to give you a free assay kit to prove it!
For the past decade scientists have extensively used ATS secondary toxin conjugates to make their own targeted toxins for in vitro use.The ability to combine...
10% Discount on 2 Rabbit Polyclonal Antibody Service. With over 20 years experience, SDIX has developed into the premier US custom antibody producer,...
Bulk Cytokines with Custom Vialing.20 - 50% off cytokines, growth factors, chemokines and more...For a limited time Cell Sciences is offering substantial...
Are you planning to have a customised antibody made for your research?Since 2000, Everest has been producing a catalog containing thousands of affinity...
Top suppliers
Agrisera AB
11 products
Biotrend
Biosensis
969 products
ABBIOTEC
3011 products
SDIX
1 products
Spring Bioscience
2291 products
Cell Signaling Technology
4976 products
Rockland Immunochemicals, Inc.
7592 products
Boster Immunoleader
1533 products
OriGene Technologies Inc.
5281 products
Maine Biotechnology Services
227 products
BD (Becton, Dickinson and Company)
1 products
ABNOVA CORPORATION
Randox Life Sciences
1502 products